 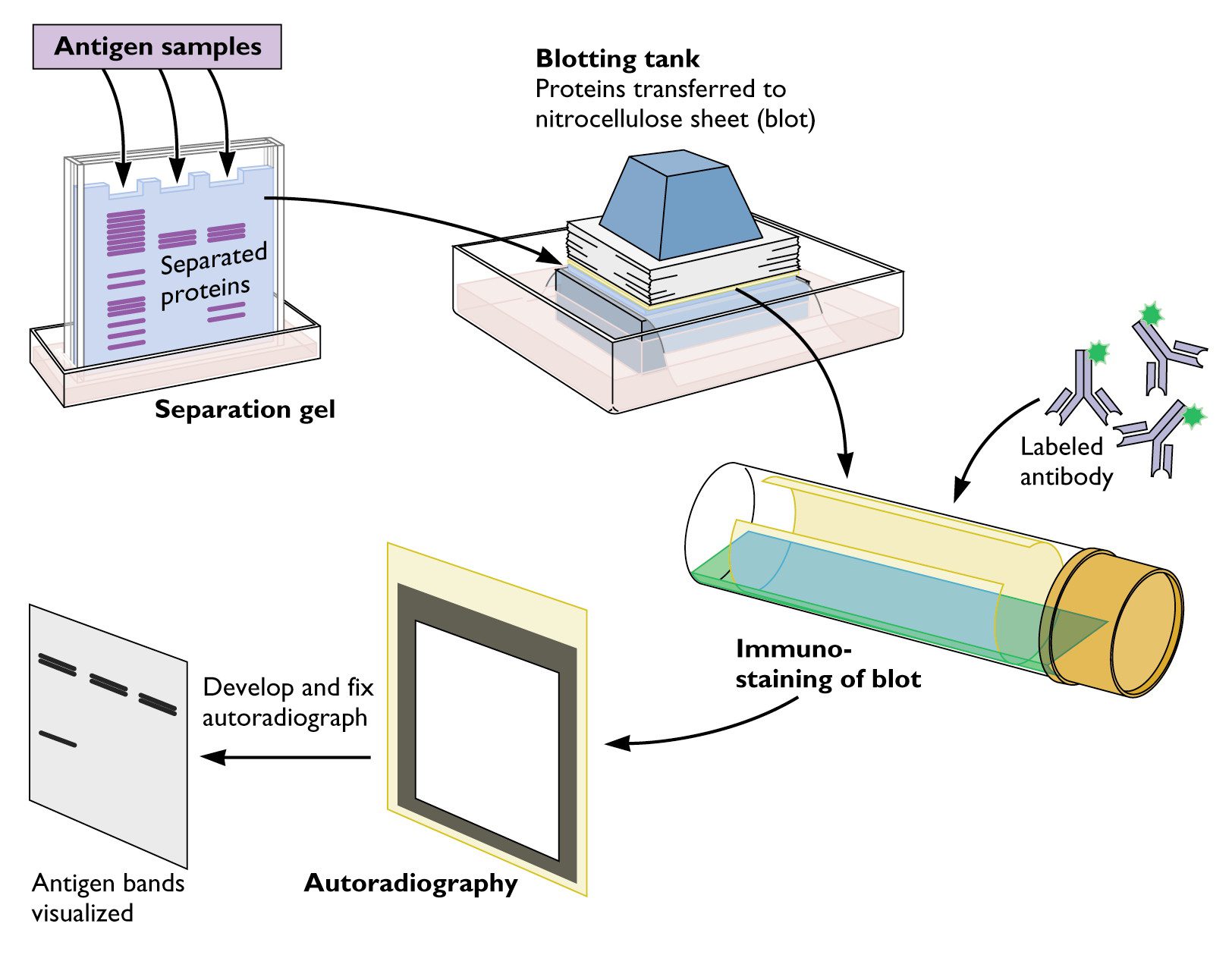
In addition, there are three visible pre-stained marker proteins at 18, 31, and 70 kD (Figure 1, right). They are detected by incubating the blot with Europium-labeled streptavidin and they show as light colored bands when read with the ScanLater Western Blot Detection Cartridge (Figure 1, left). The ScanLater Western Blot Protein Ladder consists of seven biotinylated recombinant proteins at 10, 20, 40, 50, 80, 100 and 140 kD. The blot is read with the ScanLater ™ Western Blot Detection Cartridge on the SpectraMax ® i3 or SpectraMax ® Paradigm ® Multi-Mode Microplate Readers. Detection of the biotinylated proteins is enabled by adding Europium-labeled streptavidin to the blot at the secondary antibody step. The S canLater Western Blot Protein Ladder consists of seven biotinylated recombinant proteins as well as three prestained marker proteins. The primary applications of this protein ladder include the estimation of protein molecular weight, the visualization of gel electrophoresis, and the evaluation of the transfer process from gel to blot. The ScanLater ™ Western Blot Protein Ladder is an essential component of the ScanLater ™ Western Blot Detection System. Molecular weights of detected proteins can be estimated.A single set of standards contains markers for both visual gel and blot alignment plus TRF detection.Wash excess detection reagent from the membrane before exposure.Application Note Estimation of Protein Molecular Weights with ScanLater Western Blot Protein Ladder This problem can be caused by antibodies binding to the blocking reagent in the blocking buffer. Uneven bands can be due to insufficient buffer being added to the tank during running. Uneven gel composition (gel has set too quickly while casting or buffer was mixed inadequately). Check for even protein loading by stripping and reprobing the blot with an internal control antibody (or use an HRP-conjugated loading control antibody). Uneven protein loading: assay protein samples and load by protein amount. High protein concentrations can result in diffuse protein bands. Can be caused by improper/uneven transfer or air bubbles.Reduce the voltage applied to run the SDS-PAGE gel or run the gel in a cold room. Migration through the gel was too hot or too fast. Add a blank lane between the MW marker and the first sample lane.Too much primary and/or too much secondary antibody. Inverse staining (i.e., white bands on a dark blot) Decrease the concentrations of the primary and/or secondary antibodies.Decrease the total protein loading for each sample.This happens when the ECL substrate is used up too rapidly.Try different detection reagent compositions and/or brands.ĭilute chemiluminescent reagents in high-purity water. The presence of sodium azide inhibits the activity of HRP.įilm exposure too short / detection reagent not sensitive enoughĬheck several exposure times to achieve optimum detection. Wet transfer produces higher-resolution transfers over semidry transfer. Thicker gels can result in incomplete transfer of high molecular-weight-proteins.Ĭheck the quality of protein transfer with a reversible, universal protein stain, e.g., Ponceau-S. 
Wet transfer is recommended for small proteins (10kDa).Įnsure proper transfer set-up (e.g., no air bubbles trapped between the gel and the membrane). Reduce transfer times and/or use smaller pore size membranes (0.22 μm) for low MW proteins <30kDa. Use a Tris-tricine gel for protein targets <20kDa. Switch to using an alternative blocking reagent. Reduce the percentage of, or remove, the blocking reagent from the antibody incubation buffers. Nitrocellulose works better with hydrophobic/ non-polar antigens.īlocking for too long can mask specific epitopes and prevent antibody binding. PVDF membrane works better with hydrophilic/ polar/charged target antigens. Select PVDF or NC membranes based on hydrophobicity/ hydrophilicity of the target antigen.Ĭheck the hydrophobicity/hydrophilicity of the antigen sequence. Use the optimum lysis buffer for the target protein’s subcellular localization.Ĭheck protein loading with an internal loading control antibody. Include protease inhibitors in the lysis buffer. Use appropriate treatment to induce target protein expression or modification. Adjust protein loading accordingly).Ĭhange incubation time and temperature (4☌, overnight).Įnrich low-abundance proteins by immunoprecipitation, fractionation, etc. Include a positive control (e.g., overexpressed protein, purified protein, positive cell line, etc. 
The antibody may have lost activity – perform a dot blot to determine activity and optimal concentration. Titrate the antibody to determine optimum concentration. Issues with the primary and / or secondary antibody Overcome your Western blot difficulties with our troubleshooting advice, covering problems such as weak/no signal, non-specific bands, high signal, and other common issues. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
